
Highlights
- Close collaborations with leading structural modeling systems including Rosetta (UNC, UW), Molprobity (Duke), and Phenix (Duke, Cambridge, Berkeley)
- Large scale refactoring of hydrogen bond modeling in Rosetta to accommodate higher accuracy parametrizations
- Exploration of large experimental structural biology datasets for geometric feature statistics
The Challenge
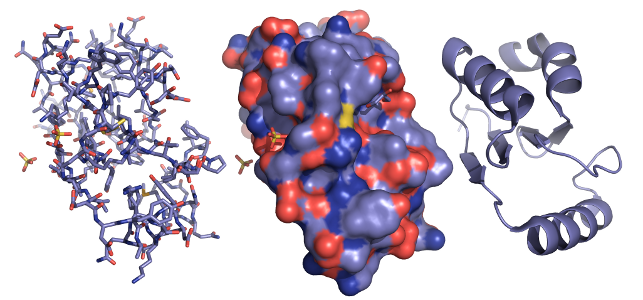
Diverse biological function is created by encoding the 3D atomic structure of macro-molecules such as Proteins and RNA in the 1D genetic sequence. Understanding the sequence to structure to function relationships allows biochemists to predict the activity of genes and rationally design genes with novel biological function.The confluence of the genomics and computer revolutions have led to new technologies that produce exponentially growing amounts of molecular structure data. Whole genomes are being sequenced, the Protein Databank has over 60,000 experimentally solved structures at atomic level accuracy and structure prediction software are generating large numbers of candidate structures on powerful compute clusters. The challenge of turning this data into knowledge is to bring together computational and biochemical thinking to develop models that are able to predict protein structure and solve important problems in biology.
The Approach
Computational models in structural biology are often surprisingly geometric: Atoms are represented as points in space and chemical bonds are represented as constraints on distances and angles between atoms. With this interpretation the structure of a macromolecule is an articulated body in a high dimensional configuration space. We can apply computational geometry techniques drawn from robotic motion planning, graphical information systems, and combinatorial optimization to build robust and scalable predictive models of molecular structure.Hydrogen Bonding
A focus of our group is building and evaluating models of hydrogen bonding with our collaborators. How do the geometric configurations of hydrogen bonds in predicted molecular structures differ from naturally occurring structures? To answer this question we are developing tools to explore and analyze ensembles of geometries. We then incorporate our findings to improve leading structure prediction and evaluation tools such as Rosetta and Molprobity.Accessibility Radius
We leverage the tools and methodology we develop in improving the modeling of hydrogen bond geometries to identify new computational geometry flavored observables of biological molecules such as the accessibility radius:The strength of a hydrogen bond has been show to depends upon its proximity to polar solvents such as water. We developed an approach to rapidly identify which bonds are accessible to water molecules by using a generalization of the Voronoi Diagram called the Power Diagram. If used in structure prediction software, the quality of a bond can be modulated to more accurately evaluate subtle properties of the hydrogen bonding interaction.
Project Leaders
- Jack Snoeyink, Professor
- Andrew Leaver-Fay, Research Professor
Graduate Research Students
- Matthew O'Meara, UNC Chapel Hill
- Vishal Verma, UNC Chapel Hill
Research Collaborations
- Kuhlman Lab, UNC Chapel Hill, Department of Biochemistry
- Baker Lab, UW Seattle, Department of Biochemistry
- Richardson Lab, Duke University, Department of Biochemistry
Selected Publications Research Sponsors NIH / DARPA